
Biosensors
Our engineering efforts aim to establish bioprocesses that maximize the production of targeted molecules while limiting fluxes to pathways that are no longer needed in an industrial setting. Measuring levels of metabolites that are internal or external to microbes involves engineering responsive elements that result in a signal — often a fluorescent one — indicative of the amount of a metabolite both noninvasively and in real time. Various biosensor strategies used in the Agile BioFoundry are highlighted here.
In vivo sensors
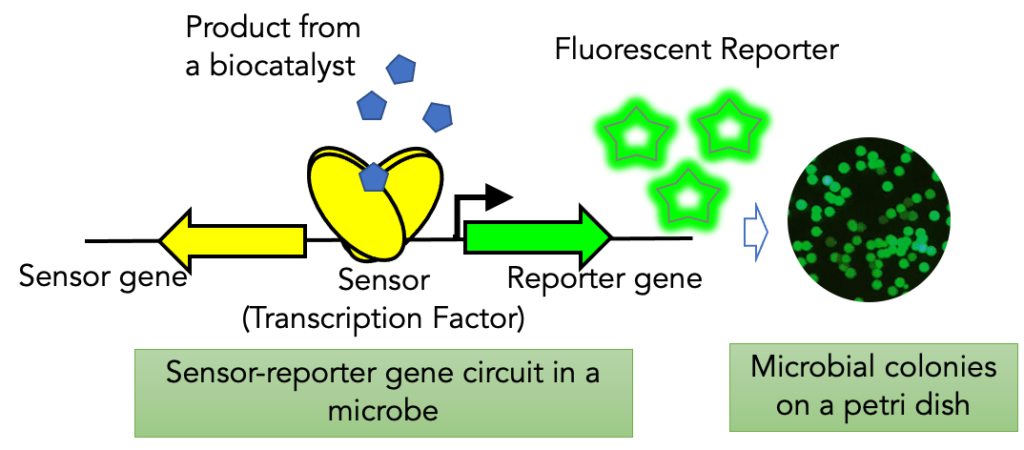
‘Smart’ microbial cell biosensor technology
While the Design and Build steps of the DBTL cycle can be high throughput, the Test step often remains a bottleneck due to the low efficiency of current screening technologies. We have developed a Smart Microbial Cell technology that uses a custom-designed sensor-reporter system to screen for a given small molecule of interest (value-added chemical, industrial precursor) in the host strain harboring variations in a single gene, or a pathway, or at the genome level. This capability allows us to incorporate computational protein design to engineer sensors or intelligent library design for the evolution of enzymes. The existing capability enhances the efficiency of the Test step of DBTL cycle for bio-based products. This technology was a recipient of a 2020 R&D 100 Award.
Transcription-factor based, fluorescent biosensors
Transcription factors are proteins that can bind to a small molecule or a metabolite and regulate the expression of a gene or a gene cluster. In the Agile BioFoundry, we are exploiting such transcription factors for biosensors to “sense” and “report” accumulation of the target molecules, which are products of bioconversion. The fluorescence signal generated from the biosensor correlates with the bioconversion efficiency and intracellular pool of the product and can be a convenient readout for titer, productivity and yield. The transcription factors are specific and tunable to have a wide range of detection to match the productivity range of engineered applied to screening of a population of biocatalyst solely based on fluorescence. These biosensors form the basis for Smart Microbial Cell Technology and can be coupled to high throughput screening efficiency of flow cytometry.
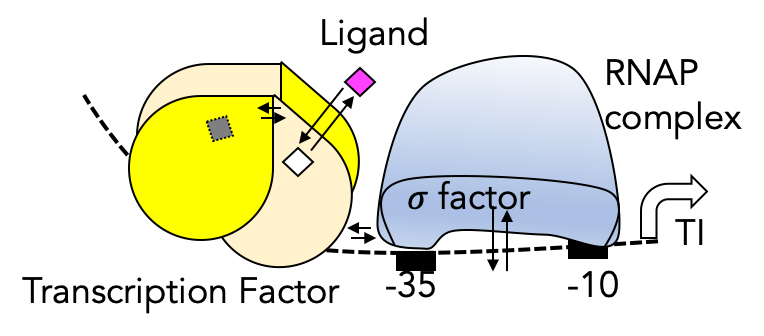
Natural transcription factors directly ported to a host organism show weak response. We have established a workflow to tune several hotspots in promoter/operator regions and in the transcription factor for an optimal response to the target molecule.
In vitro sensors
FRET biosensors
Fluorescence resonance energy transfer (FRET)-based biosensors couple ligand-dependent protein conformational changes to a ratiometric fluorescent signal response. Such biosensors can be used intra- or extracellularly to provide rapid, real-time responses to ligands or metabolites of interest.
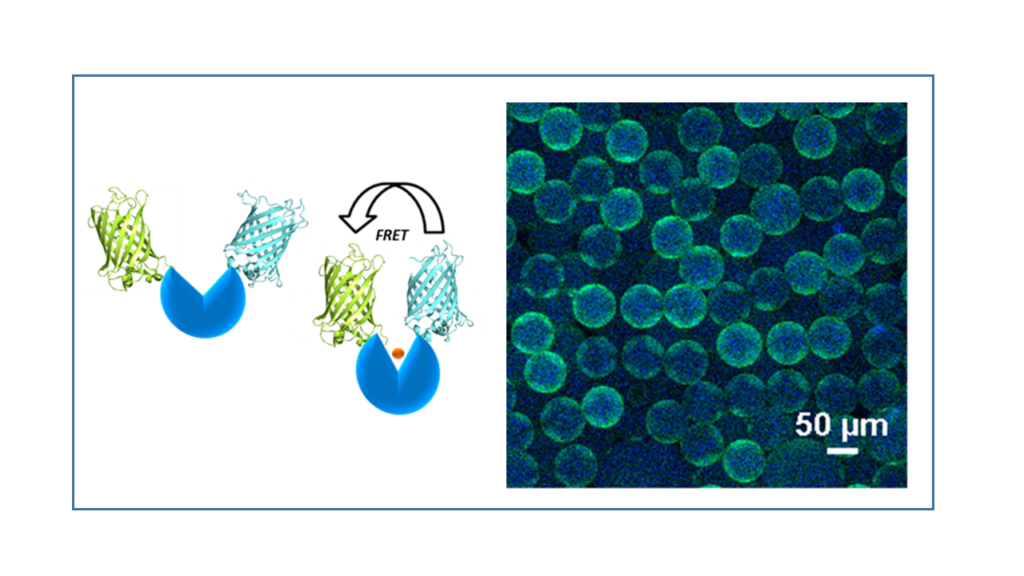
We can design and optimize FRET biosensors for a variety of target molecules using an in-house library of fluorescent protein scaffolds.
Reporter-cell-based biosensors
Reporter-cell-based sensors are capable of measuring bioproduct or feedstock from both. Introducing these biosensors into the production host can be used to select good performers via fluorescence-activated cell sorting.