
Biosensors
Custom-made biosensors to detect intracellular metabolite concentration and enhance throughput
Biosensors are molecular elements used to noninvasively measure intra-, or extra-cellular levels of metabolites in real time. ABF has developed tunable biosensors for monitoring the production of several molecules of interest. Our biosensors are able to distinguish between small differences in concentration, at relevant concentration ranges.
In vivo sensors
‘Smart’ microbial cell biosensor technology
Our Smart Microbial Cell Technology is a high throughput screening platform for enzymes and whole cell biocatalysts. It uses a custom-designed sensor-reporter system to screen for a given small molecule of interest (such as a value-added chemical or industrial precursor) in the host strain harboring variations at a single gene, pathway, or genome level. This disruptive technology allows rapid screening of large libraries to capture rare gain-of-function events that result in improved enzymes and biocatalysts. Learn more about the Smart Microbial Biosensor Technology and watch a video about this technology.
Transcription-factor based, fluorescent biosensors
Transcription factors are proteins that can bind to small molecules or metabolites and regulate gene expression. We use transcription factors for biosensors to sense and report the accumulation of target molecules. The biosensor generates a fluorescent signal that correlates with bioconversion efficiency and the amount of product inside the cell, making it easy to measure titer, and calculate productivity and yield.
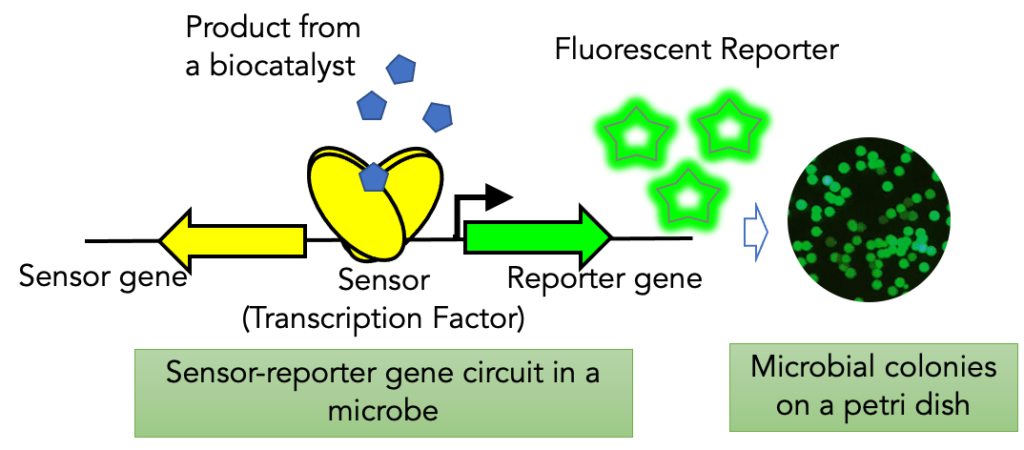
Natural transcription factors directly ported to a host organism show weak response. We have established a workflow to tune several hotspots in promoter/operator regions and in the transcription factor for an optimal response to the target molecule.
In vitro sensors
FRET biosensors
Fluorescence resonance energy transfer (FRET)-based biosensors couple ligand-dependent protein conformational changes to a ratiometric fluorescent signal response. These biosensors can be used intra- or extracellularly to provide rapid, real-time responses to ligands or metabolites of interest. We can design and optimize FRET biosensors for a variety of target molecules using our in-house library of fluorescent protein scaffolds.
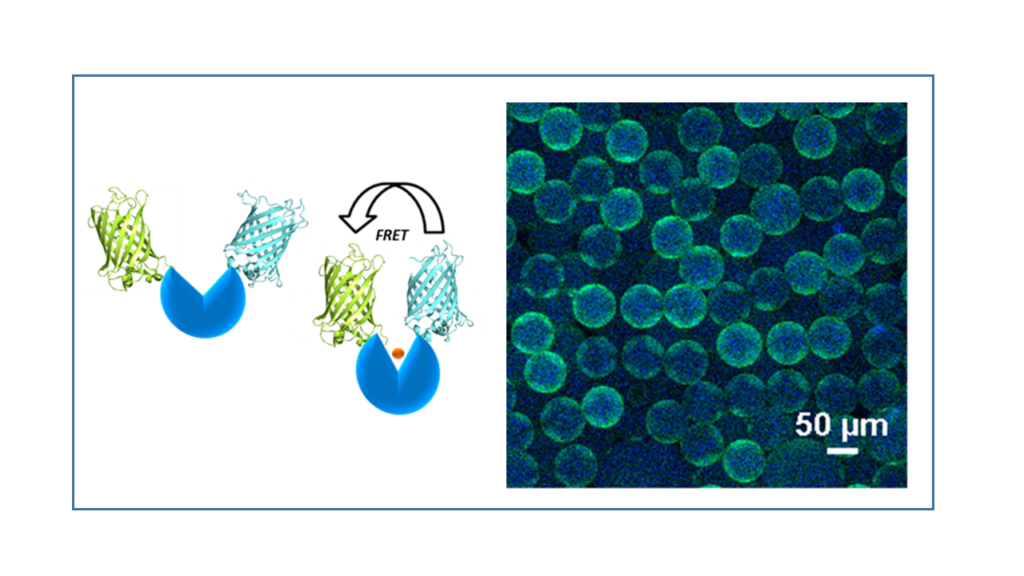
Reporter-cell-based biosensors
Reporter-cell-based sensors are capable of measuring bioproduct or feedstock. These biosensors can be introduced into production hosts to select good performers via fluorescence-activated cell sorting.
USE CASE
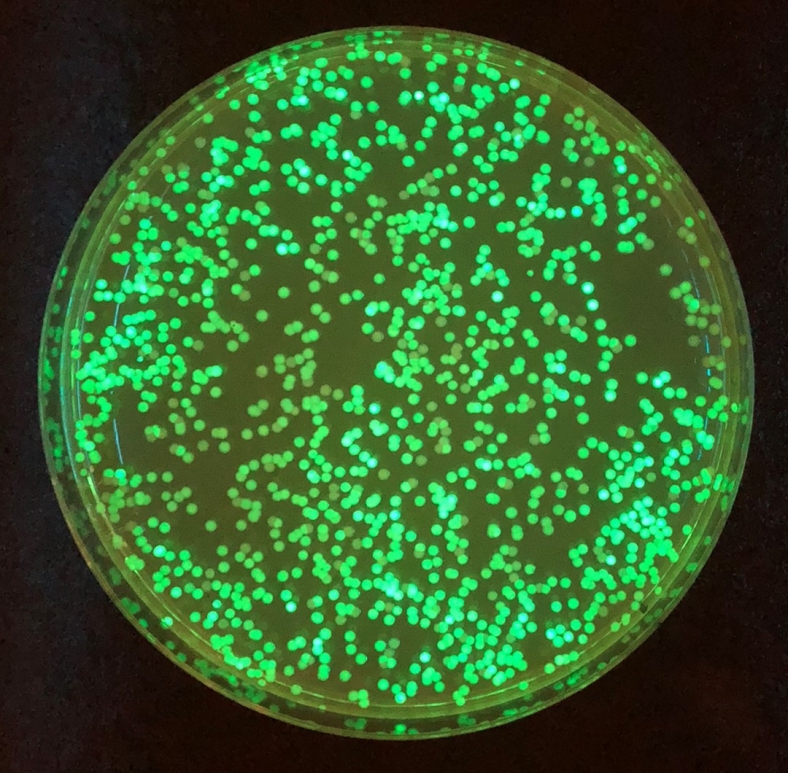
Agile BioFoundry researchers developed a fluorescent biosensor that confirmed the transport of terephthalic acid in the soil bacterium Acinetobacter baylyi ADP1.
This confirmed bioproduction of terephthalic acid as well as successful degradation of poly-ethylene terephthalate into terephthalic acid.
Publications
- Biosensors for the detection of chorismate and cis,cis-muconic acid in Corynebacterium glutamicum
- Tackling the Catch-22 Situation of Optimizing a Sensor and a Transporter System in a Whole-Cell Microbial Biosensor Design for an Anthropogenic Small Molecule
- Gene amplification, laboratory evolution, and biosensor screening reveal MucK as a terephthalic acid transporter in Acinetobacter baylyi ADP1
- Engineering glucose metabolism for enhanced muconic acid production in Pseudomonas putida KT2440